European (EU) H2020 funded VANGUARD project is set to generate a tissue engineered bioartificial pancreas, which, due to its high translation potential, meets all the criteria for the next stage from bench to bedside. The developed beta cell replacement therapy will be available to larger numbers of type 1 diabetic patients.
VANGUARD
Brief Description & Goal
Coordinated by University of Geneva a multidisciplinary team of researchers will develop a clinical beta-cell replacement therapy and overcome the limitations of current therapies. As a consequence, efficacy and patient safety can be drastically improved. Type 1 diabetes is the most common chronic disease in children and adolescents. Despite primary treatment with exogenous insulin therapy, the blood sugar level cannot be perfectly controlled and has a negative impact on quality of life. In addition, chronic complications can occur. At this moment, the treatment option is a transplantation of either the entire pancreas or isolated islets, but there is a marked imbalance between organ donors and the people suffering from the disease. Additionally, a lifelong anti-rejection medication to avoid the rejection of the transplanted organ or cells is required.
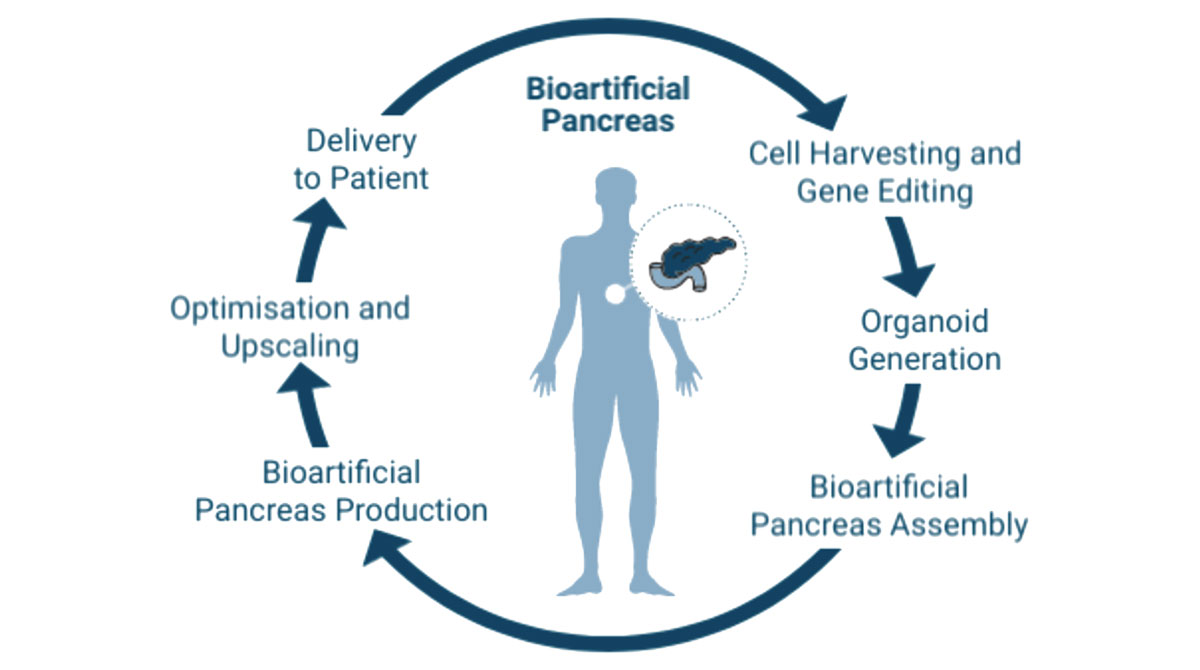
The bioartificial pancreas offers a breakthrough in the field of regenerative medicine and will be used as an Advanced Therapy Medicinal Product (ATMP) and a cell-based treatment for type 1 diabetes. Therefore, insulin-producing organoids are integrated into a functionalized hydrogel. The existing extracellular matrix of the construct has anti-inflammatory and immunomodulatory properties and promotes vascularization.
The aim of the project is to generate a vascularized and immune-protected bioartificial pancreas as a beta cell replacement therapy to improve the treatment of type 1 diabetes.
Our Role
Kugelmeiers Ltd. has long-standing experience in the field of scaffold-free 3D cell culture and the production, handling and cultivation of (stem cell-based) spheroids. With the Sphericalplate 5D, the company has developed a translational platform for upscaling of standardized spheroids of any type and size. Kugelmeiers will lead the pre-clinical development, support the Ethics, Legal and Social Aspects (ELSA) and project partners as Innovation Manager. In addition, an internal team of regulatory specialists is accompanying the project in order to prepare the approval of the developed medical devices and cell products as a clinical therapy for the treatment of Type 1 diabetes.
Link project website: www.vanguard-project.eu
Project Partners
| University of Geneva, Department of Surgery (Project coordinator) www.unige.ch Switzerland |
The School of Medicine of Università del Piemonte Orientale “Amedeo Avogadro” www.diss.uniupo.it Italy |
The Transplantation department of Lyon University Hospital www.chu-lyon.fr France |
| IRCCS Ospedale San Raffaele, Diabetes Research Institute www.research.hsr.it Italy |
Ludwig-Maximilians-Universität München, Diabetes Center www.klinikum.uni-muenchen.de Germany |
Erasmus University Medical Center Rotterdam www.erasmusmc.nl The Netherlands |
| Accelopment AG (Project Management) www.accelopment.com Switzerland |
European Society for Organ Transplantation, Ethical, Legal and Psychosocial Aspects of organ Transplantation (ESOT-ELPAT) www.esot.org Italy |



